Abstract
Background: Regulatory T-cells (Tregs) are a T-cell subset capable of suppressing effector T-cells (Teffs) and, when given at the time of allogeneic hematopoietic stem cell transplantation (Allo-SCT), have been shown to prevent graft v. host disease (GVHD). We previously demonstrated that cord blood (CB) Tregs can be ex vivo expanded to clinically meaningful doses and prevent GVHD in a xenogenic mouse model. The efficacy of the ex vivo expanded CB Tregs in preventing GVHD was markedly improved with fucosylation, which adds fucose resulting in creation of a siayl-Lewis X moiety found on P-selectin ligand, thereby facilitating trafficking through the endothelium into inflammatory sites. We hypothesized that adoptive therapy with fucosylated Tregs could prevent GVHD even when given at suboptimal Tregs:Teffs ratios. We conducted a Phase I clinical trial to evaluate the clinical safety of fucosylated CB Treg infusion for GVHD prophylaxis (ww.clinicaltrials.gov NCT02423915). We now report on 5 patients who received GVHD prophylaxis with 3rdparty, ex-vivo expanded CB Tregs at a dose level of 1.0 x10e6 cells/ kg (2 un-fucosylated and 3 fucosylated CB Treg products).
Patients and methods: A 3rd party CB unit with at least 3/6 HLA matching to the recipient was selected. Treg isolation and expansion was performed as shown previously. When indicated, expanded CB Tregs were incubated with substrate (GDP β-fucose) and fucosyltrasferase-VI (FT6) enzyme (TZ101, Targazyme, Carlsbad, CA) for 30 minutes, washed and infused on day -1. The first 2 patients underwent double cord transplant with Flu/Cy/TBI reduced intensity conditioning and received unfucosylated CB Tregs at a dose of 1 x 10e6/kg. The subsequent 3 patients underwent peripheral blood (PB) matched unrelated donor (MUD) transplant with Fludarabine/ Melphalan myeloablative conditioining and received fucosylated CB Tregs at a dose of 1 x 10e6/kg. All patients received additional pharmacologic GVHD prophylaxis with sirolimus and mycophenolate mofetil. All patients received their designated CB Treg dose. The median proportion of CD4+25+CD127- in the infused Treg product was 96% (94-98%). Expanded CB Treg product showed suppression of T cell proliferation using CFSE assay. For the first 2 patients, the infused graft Teffs dose was at least 12 times more than the CB Tregs and for the subsequent 3 patients, the dose of infused Teffs dose was at least 132 times more than that of the infused fucosylated CB Tregs. No infusion toxicities were observed.
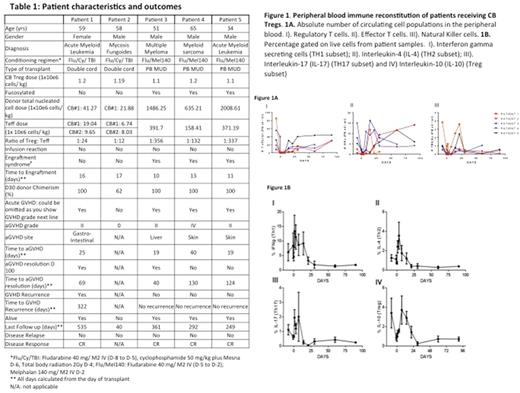
Results: All patients receiving fucosylated CB Tregs followed by PB MUD transplants exhibited non-infectious high fevers up to 40οC accompanied by an erythematous rash starting on post-transplant day +5 that lasted for 5-7 days and resolved after steroid administration (IV methylprednisolone 1mg/kg) for 24-96 hours. Skin biopsies were consistent with engraftment syndrome and not GVHD. All 5 patients engrafted at a median of 13 days. The three patients who received fucosylated CB Tregs and one of two patients receiving untreated CB Tregs developed ≥ grade II acute GVHD at a median of day +22 which had completely resolved by day 130 post-transplant. One patient had late onset aGVHD. No cGVHD was documented in any patient. One recipient of non-fucosylated Tregs died on day +40 due to unrelated causes (intracranial bleeding). No GVHD was observed in this patient. PB Flow cytometric analysis revealed an increase in inflammatory T-cell subsets in the first week post-transplant with a concurrent bimodal increase in the Treg populations on day -1 and day +7 (figure 1A). NK and DC but not B cell subsets were significantly increased. A consistent increase in the cell populations secreting IFNү, IL-4 and IL-17 was observed in the post-transplant period and a bi-modal increase in the IL-10 secreting cells was observed on Day -1 and Day +7 consistent with the circulating Treg cells (figure 1B).
Conclusions: Our group is the first to show safety of infusing 3rd party, ex-vivo expanded CB Tregs in a PB MUD transplant setting. At a dose level of 1.0 x 10e6 /kg, 3rd party CB Tregs were administered without acute infusional toxicity and no negative impact on engraftment. The cause for the high fevers observed in the fucosylated CB Treg recipients is unclear at this time. We are now enrolling additional patients with unfucosylated Tregs in a PB MUD transplant setting to investigate the mechanisms underlying this phenomenon.
Khoury: Kiromics: Research Funding; Angle: Research Funding; Stemline Therapeutics: Research Funding; Pfizer: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.